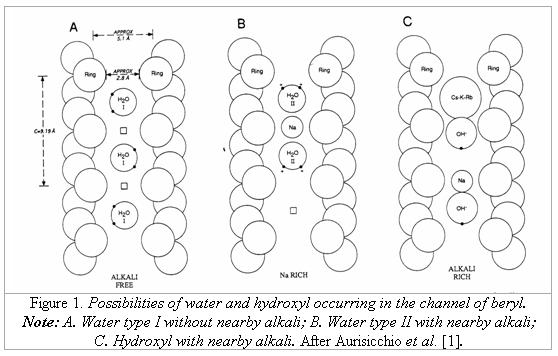
CLASSIFICATION
OF WATER TYPES IN STRUCTURAL CHANNEL OF BERYL BY MEANS OF
RAMAN SPECTROSCOPY
LÊ THỊ
THU HƯƠNG1,2, WOLFGANG HOFMEISTER
2
1Faculty of Geology, Hà
Nội University of Sciences, Hà Nội, Việt Nam
2Centre of Gemstone Research, Johannes Gutenberg University, D-55099
Mainz, Germany
Abstract: Two types
of water molecules in the structural channel of beryl are classified by Raman
spectroscopy under room temperature. The type I is that occuring alone and
with a typical orientation in the way that the symmetry axis of water molecules
is perpendicular to the c axis of the emerald crystal; the type II is water
molecules which are associated with nearby alkalis and the water molecule
symmetry axis is parallel to c axis of the host crystal as the result of
interaction with alkali ions. Under low temperatures (198-123o K) the splitting of the band of water type
II is present and possible reasons for the band splitting are discussed.
I. INTRODUCTION
The types of water molecules in structural channels of beryl (Be3Al2Si6O18) have been classified by infrared spectroscopic studies in mixed powdery beryl and KBr pellets since more than 40 years ago [6, 7]. This article brings out the possibility of classifying water types by non-destructive Raman spectroscopy. As related to alkali amount, water molecules exist differently in type and “density” in beryl of different origins that can be detected by Raman spectroscopy, and since the method does not require taking material from samples it could be very useful in the determination of gem origin.
According to Wood & Nassau [6, 7], the water of type I is that which occurs alone and with a typical
orientation in the way that the symmetry axis of the water molecule is perpendicular
to the c axis of the emerald crystal; the type II is a water molecule which is
associated with nearby alkalis, and the water molecule symmetry axis is
parallel to c axis of the host crystal as the result of interaction with alkali
ions. In their studies concerning the distinction of natural and synthetic
emeralds, Schmetzer [4] and Schmetzer and Kiefert [5] supplemented that water molecules can either exist alone or with nearby
alkalis (mainly sodium). In the case, that there is a nearby alkali ion, the
water type II may be associated with this ion in two sequences: H2O-Na-OH2
or H2O-Na-, in which represents the vacancy of water (no water) in the channel sites.
Therefore, they divided the water type II into two subtypes: the subtype IIa is
water molecules like H2O-Na-OH2, and the subtype
IIb is water molecules like H2O-Na-. Furthermore, they found that also hydroxyl ions (

II. MATERIAL AND EXPERIMENTAL METHODS
Single crystals of natural emeralds from
The presented Raman spectra were done in the confocal measuring
mode by the Jobin Yvon (Horiba group) LabRam HR 800 spectrometer. The system was
equipped with an Olympus BX41 optical microscope and a Si-based CCD
(charge-coupled device) detector. The spectra were excited by the Ar+
ion laser emission of 514 nm as a green laser with a grating of 1800
grooves/mm. Due to these parameters and the optical path length of the
spectrometer, a resolution of 0.8 cm-1 was achieved. The spectra acquisition time was a set of 240 seconds for all measurements.
The range of water was measured from 3500 cm-1 to 3700 cm-1
under room to low temperature (from 300 K to 78 K) in steps of 25 K. The peak
analysis was performed with an Origin-lab 7.5 professional software package.
The single or overlapping peaks were fitted using Gauss-Lorentz function.
The alkali concentrations were analyzed by LA-ICP-MS. The
ablation was achieved
with a New Wave Research UP-213 Nd:YAG laser ablation
system, using a pulse repetition rate of 10 Hz and 100 µm crater diameters.
Analyses were performed on an Agilent 7500 ce
inductively coupled plasma-mass spectrometer in pulse counting mode (one point
per peak and 10 ms dwelling time). Data reduction was carried out by the
software “Glitter”. The amount of material ablated in laser sampling is
different for each spot analysis. Consequently, the detection limits are
different for each spot and are calculated for each individual acquisition. The
detection limits generally range between 0.001 and 0.5 ppm (µg/g). 28Si
was used as the internal standard. Analyses were calibrated against the
silicate glass reference material NIST 612 using the values of Pearce et al. [3], and the US Geological Survey (USGS) glass
standard BCR-2G was measured to monitor accuracy.
III.
RESULTS AND DISCUSSION
1.
Room temperature measurements
Under room temperature,
all samples of this investigation were measured for obtaining the spectra in
the range of
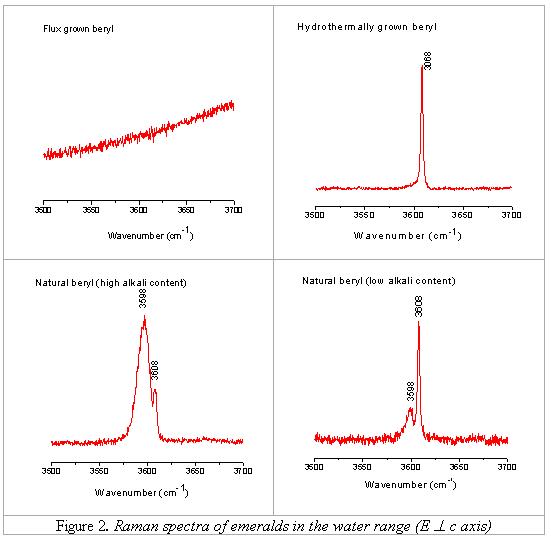
Considering chemical
data, it is obvious that the appearance of the band at 3598 cm-1 and
also the intensity ratio of the two bands (3598 and 3608 cm-1) depend
on the amount of alkali ions. The band 3598 cm-1 is detectable only
in alkali-bearing samples; in alkali-free samples (hydrothermal synthesis) this
band absolutely disappears while the band 3608 cm-1 truly exists.
This band increases in intensity as the alkali content of emerald increases,
and the more alkali ions are present in sample, the higher is the ratio I3598/I3608.
In those samples with
the amount of alkali ions higher than 1,1 wt% (emeralds from Brazil, Russia,
Austria, Madagascar, Zambia, South Africa) the intensity of the band 3598 cm-1
is higher than that of the band 3608 cm-1 (I3598/I3608
> 1), and on the contrary; in those samples with the amount of alkali ions
lower than 1.1 wt% (emeralds from Colombia, Nigeria, China and synthetic
“emeralds”) the intensity of the band 3608 cm-1 is higher than that
of the band at 3598 cm-1 (I3598/I3608 < 1)
Figure 3). According to this observation and to the classification of Wood
& Nassau [6, 7], it can be stated here, that the Raman band at 3608 cm-1
is generated by the water type-I (those water molecules having not any presence
of nearby alkali ions) and the Raman band at 3598 cm-1 is generated
by water type-II (water molecules with nearby alkali ions).
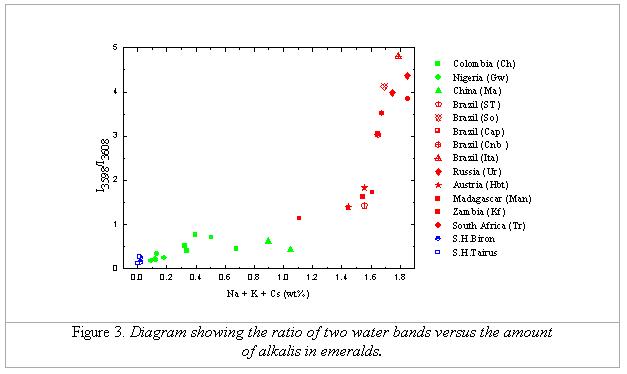
In all natural stones, the band at 3598 cm-1
(water type II) shows to be much broader than the band at 3608 cm-1
(water type I). The FWHM values of band 3598 cm-1 range from 11.2 to 14.8 cm-1 while those of band 3608 cm-1 (water type I) range from 1.6 cm-1 to 2.8 cm-1. The reason that the Raman band of water type II is broader than that
of type I may be due to the vibrational characteristics of each water type
itself. This means, since there is the appearance of nearby alkali ions (in
cases of water type II), the orientation of water molecules changes; therefore,
the vibration energy and/or state of vibration of water molecules actually
changes. This explains why the water type-II molecules generate the Raman band
at different positions and have different FWHMs than the water type-I molecules.
Nevertheless, it may be also the case that, the broadening of the band 3598 cm-1
is caused by the combination of more than one band, since its width is not
normal for a single typical Raman band. According to the study of Schmetzer
& Kiefert [5], the water type II can be subdivided into the subtype IIa (H2O-Na-OH2) and subtype IIb (H2O-Na-ÿ). Therefore, it could be supposed here, that the band of 3598 cm-1
is not a single band, but in fact a combination of at least two bands, one due
to the type-IIa water and the other due to the type-IIb.
2. Low temperature
measurements
To investigate the behaviour of this broad band, the
Raman measurements were carried out under different temperature levels. One sample from
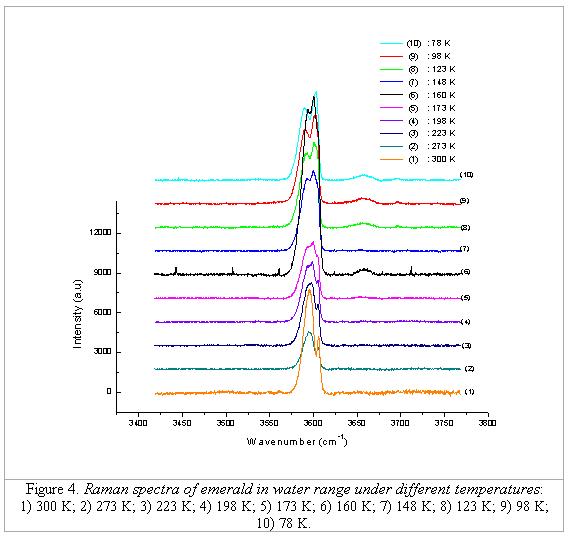
From 300 to 223 K the
spectra show to maintain 2 peaks, although the FWHM values and their position
changes under different temperatures. In addition, the lower the temperature
is, the closer the two peaks come together (from 11.6 cm-1 when
measured under room temperature, to 10.3 cm-1 when measured at 223
K). The FWHM of the band 3596 cm-1 increases,
while that of band 3608 cm-1 decreases. From 198 to 123 K, the
spectra have clearly 3 peaks (Figure 5); only lower than 198 K the splitting of
the band of the water type II are present. Below 98 K, again only two peaks are
detectable, one at 3593 cm-1 with lower intensity and another at
3604 cm-1 with higher intensity.
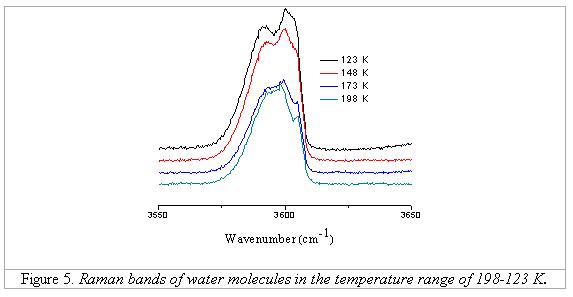
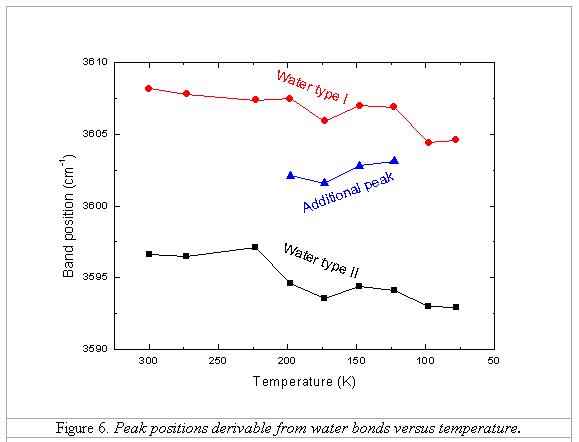
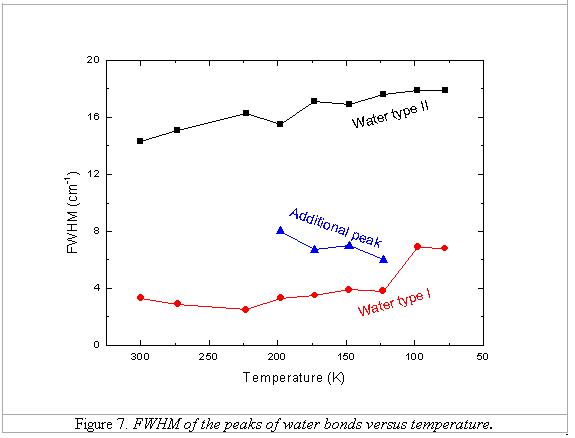
Figures 6 and 7
illustrate the diagrams showing the appearance of the additional peak between
123 and 198 K, and the change of band positions and FWHMs of two types of water
bands according to the change of temperature. There are some proposals for the
following appearance of the additional peak. However, it has to underline, that
the presented proposals need further experimental confirmation. We need a
strictly accurate polarizer filter 1. Following the study of Schmetzer &
Kiefert [5], in which 3 types of water have been assigned, the type I, type IIa
and type IIb by IR spectroscopy, the additional peak is seen in Raman
spectroscopy, therefore it could be assigned for the third type of water. And,
the peak which is detected at room temperature at 3598 cm-1, is the
overlap of two water types IIa and IIb. At low temperatures, the silicate
structure of emerald is slightly constricted. Accordingly, the deformation or reorientation of the water
type II molecules may take place (remember that the water type II molecules are
oriented in such a way that their two-fold symmetry axis is parallel to the
crystallographic six-fold axis of the emerald structure). This means, the
parallel position changes slightly to the diagonal position. This new position generates
a new energy which is recordable as an additional peak in the spectrum.
IV. CONCLUSIONS
In this article, the
types of water have been studied and classified by non-destructive Raman
spectroscopy. The type I are those molecules which occur without nearby
alkali, and the type II are those which are associated with nearby alkali ions.
The singular broadening of the band at 3598 cm-1 (water type
II) and the splitting of this band under low temperatures between 198 and 123 K suggest
that, this band may be the overlap of two subtypes of water (IIa, IIb) in the
sequences like H2O-Na-OH2 and HO-Na-. It
needs nevertheless further study to confirm if the splitting of the band of
water type II under low temperatures is caused by different sequences or simply
by the deformation and
reorientation of the water type II, as the result of a constriction of the channel
structure.
Acknowledgments: This work has
been financed by the Johannes Gutenberg - University Research Fund for Gemstone
Research. Analytical facilities have been provided by the
REFERENCES
1. Aurisicchio C., Grubessi O., Zecchini P.,
1994. Infrared spectroscopy and crystal chemistry of the beryl group.
The Canad. Mineralogist, 32 : 55-68.
2. Brown G.E., Milis B., 1986. High-temprature
structure and crystal chemistry of hydrous alkali-rich beryl from the Harding
pegmatite,
3. Pearce N.J.G., Perkins W.T., Westgate J.A., Gorton M.P., Jackson S.E.,
Neal C.R., Chenery S.P., 1997. A compilation of new and published major and
trace element data for NIST SRM 610 and NIST SRM 612 glass reference materials.
Geostandards Newsl., 21
: 115-144.
4. Schmetzer K., 1989. Types of water
in natural and synthetic emerald. Neues
Jahrb. für Miner. Monatshefte, p. 15-26.
5. Schmetzer K., Kiefert L. 1990. Water in beryl: A contribution to the
separability of natural and synthetic emeralds by infrared spectroscopy. J. of Gemmology, 22/4 : 215-223.
6. Wood D.L. and
7. Wood D.L. and